Hydrogen Bound Tropolone Complexes
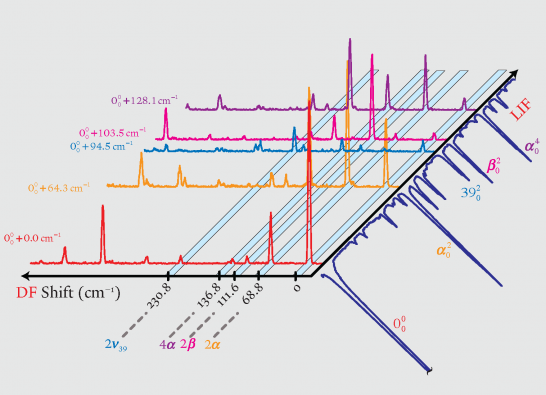
The potentially frustrated transfer of a proton between the hydroxylic (proton-donating) and ketonic (proton-accepting) oxygen atom centers in tropolone (TrOH) long has served as a model system for the study of coherent (symmetrical) proton-transfer events. A litany of hydrogen-bound complexes [TrOH • Xn] can be formed in situ by docking amphoteric ligands onto the TrOH substrate under supersonic free-jet expansion conditions. Binary (n = 1) and higher order (n = 2, 3, . . .) complexes formed with formic acid, hydrogen fluoride, acetic acid and propiolic acid (X = FA, HF, AA, and PA) have been synthesized and interrogated using a variety of spectroscopic probes built upon the intense Ã1B2 - X̃1A1 (π*←π) near-ultraviolet absorption system of bare tropolone, thereby providing vibronically resolved information through combined use of laser-induced fluorescence (LIF), dispersed fluorescence (DF), fluorescence hole-burning (FHB), and stimulated emission pumping (SEP) methods. Experimental results reveal the propensity for binary complexes to adopt a higher-energy external binding motif (ligand attached to the seven membered aromatic ring) over the energetically preferred internal form (ligand bound to the O — H ••• O reaction center), where the latter cleft-bound species can undergo unique symmetric (coherent) double proton-transfer reactions.